Orthopaedic Implant Wear Testing & Tribology Services
Our orthopaedic wear and fatigue Center of Excellence (CoE) was designed to offer support to Original Equipment Manufacturers (OEMs). Our CoE is equipped with all the testing rigs and analytical instruments needed to produce the data our clients need - but it is our dedicated team of expert scientists and engineers that really supply the added value.
With over a hundred years of experience spread throughout our team, you gain access to orthopaedic engineers, regulatory specialists, analytical chemists, polymer scientists, metallurgists, dedicated wear simulation experts, and powders experts. Not to mention the ceramics expertise that our company was founded upon over 100 years ago, an area of expertise that is still present in our advanced ceramics offering today.
If you are looking to:
- Simulate and evaluate the wear performance of your new bearing materials
- Compare an innovative design with an existing one
- Outsource your orthopaedic implant wear testing to accelerate new product developments and avoid in-house delays
- Find a supplier that can supply a comprehensive service and the full suite of information you require for your regulatory submission
- Find out what is happening at the surface of your material
...then we can help.
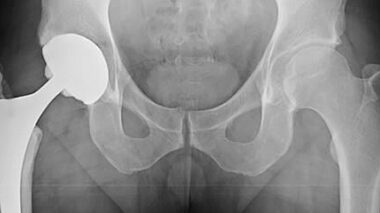
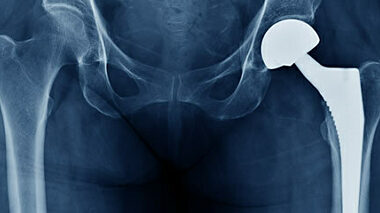
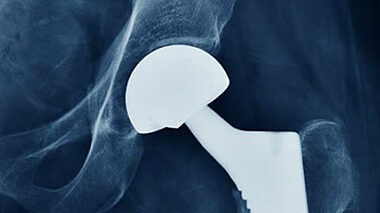
We offer wear testing and tribology, plus supporting services, for hip, knee, shoulder, spine, ankle, and other implants.
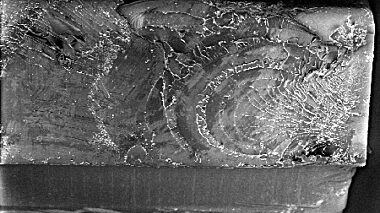
Debris Analysis
Using a combination of techniques, we offer analysis of debris generated through wear testing. In addition to following the ASTM standard, using SEM (Scanning Electron Microscope) evaluation to supply qualitative information, we also use laser diffraction techniques to gather more detailed, quantitative information, such as chemical analysis to understand the composition of the particles and particle size distribution.
Surface Analysis
We supply real understanding of what is happening at the interface between your materials using our advanced surface science techniques and expert analysis.
- Does a coating or process used to improve the hardness / wear properties of your component affect the base material in any way?
- Does the articulation cause material transfer or migration across the interface or into the surface that could be detrimental?
- Is a chemical reaction occurring between the dissimilar materials and affecting the properties or biocompatibility of the components?
- Is the wear pattern consistent and, if not, is it because of the materials or the design?
White Papers