Orthopaedic Testing Services
From an initial service offering of wear testing of orthopaedic implants, Lucideon has expanded our service portfolio within this area of Healthcare to include:
- 510k approvals
- Cleaning, sterilisation, and validation
- Failure analysis
- Regulatory strategy testing (including biocompatibility testing)
- Static and fatigue testing
- Wear testing and tribology
All the work we conduct is performed with expertise, and we will always go the extra mile to ensure that our results are of the very highest standard. We offer consultancy with experts for all the testing that we do, and if you need further interpretation of results, we can provide the expertise.
Orthopaedic products and materials sometimes require other testing facilities to determine parameters that are out of scope of standard tests.
Lucideon has extensive testing facilities to determine any non-standard parameters you may need to consider, such as:
- Trace analyses by ICP-MS (Inductively Coupled Plasma – Mass Spectrometry)
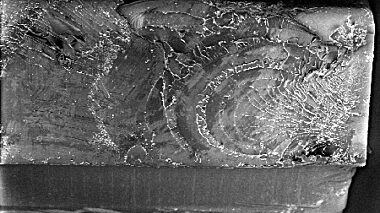
- Microstructural imaging (including contamination analysis) by SEM and FEG-SEM (Scanning Electron Microscopy and Field Emission Gun Scanning Electron Microscopy)
- Stress and strain measurements by DIC (Digital Image Correlation)
White Papers