Case Studies
At Lucideon, we have worked on many different and diverse projects across a wide range of materials and industries. Read here about some of the work we have done

Application of Lucideon's Flash Sintering Technology

Testing carried out at Lucideon's GMP testing laboratories and analytical chemistry facilities
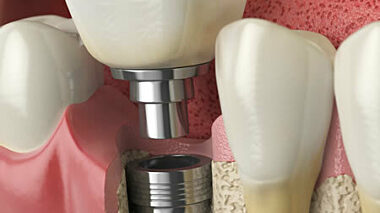
Characterisation of the surface chemical composition of medical implants

A high-resolution surface topography method was utilised to measure dentine tissue loss and surface roughness

Testing provided evidence that the strength requirements of the UHPFRC were met at each stage of the flyover restoration process

One of Mauer’s unique EWI systems achieved certification through the development of a customized testing program

Lucideon testing helps to optimize the coating process of powders

Lucideon's additive manufacturing expertise helped identify root cause of product failure

Monitoring of the in-depth distribution of key alloying elements and trace elements using SIMS

Lucideon assists in identification and prevention of staining of orthopaedic instruments

IKEA wished to appoint a preferred consultant for ceramic product sourcing around the globe

Brickwork testing for the Battersea Power Station redevelopment project

Testing enabled Hemsec to attain ETA accreditation and CE certification for their structural insulated panels

Testing of unique cladding system and its components

Benchmarking study carried out by Lucideon
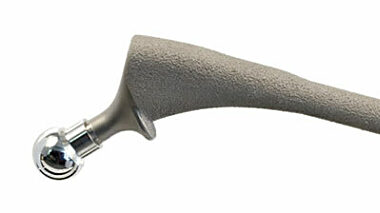
Surface contamination investigation

Lucideon has worked with Greatbatch Medical to help them gain FDA approval
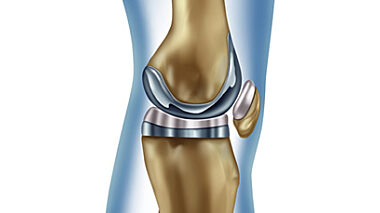
Testing and analysis provided by Lucideon

A customised materials testing project for Nuclear Transport Services (NTS), UK (Phase 2)

A customised materials testing project for Nuclear Transport Services (NTS), UK (Phase 1)

Environmental monitoring qualification of new facilities

Resolution of a manufacturing issue with fragile tableted products

Validation of the effectiveness of detergents, cleaning wipes and spray and wipe products in removing surface bacteria
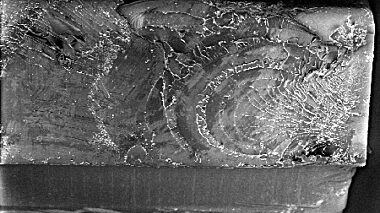
Analysis of failed polymer components in orthopaedic instruments using a range of testing and analysis techniques

Analysis of concrete and screed identified that the flooring materials specified were not used and lower-cost alternatives were sourced

Testing and consultancy enabled third party insurance and building control sign-offs
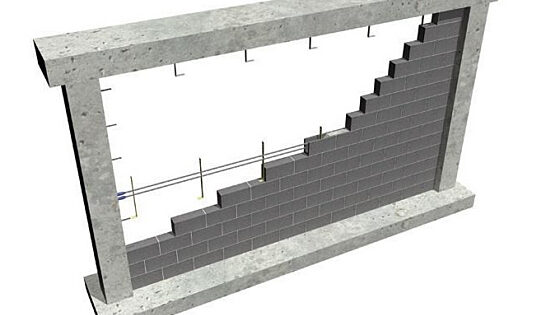
Tests to determine the strength of walls constructed with bond beams
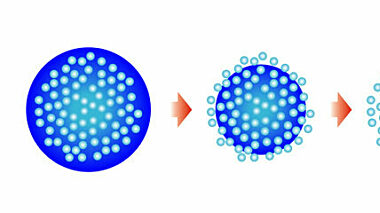
Encapsulation of bleach helps to maintain activity in application environment

Lucideon method development helps quantify effectiveness of washing machine soap

Lucideon's additive manufacturing expertise helped identify root cause of product failure

Lucideon expertise helped a client with additive manufactured implants

We helped our client with discolouration issues after hot isostatic pressing
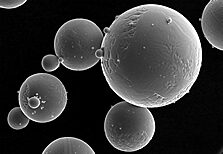
Lucideon expertise helps develop powder conditioning procedure to improve processing
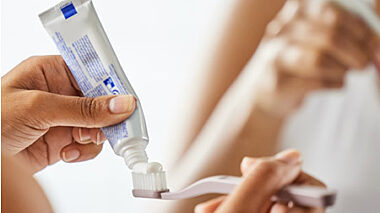
Lucideon testing helps to prove effects on tubule occlusion

Lucideon expertise allows new elements to be included in products.
Lucideon helps in product performance comparison
Optimisation of powders for tablet pressing

Lucideon uses surface analysis to investigate hair care products

Topographical analysis of skin

Application of Lucideon's cleaning validation procedure - Validata

New standards produced for Euro currency colour consistency

Standards to ensure consistency in the calibration of the instruments used to measure raw wool colour

A manufacturer of ceramic armour for military vehicles and personnel sought pressure-cast forming expertise
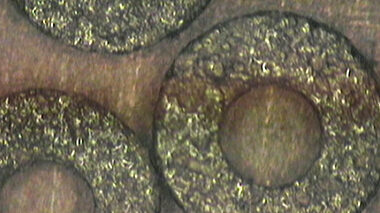
Testing and reverse engineering of materials

ToF-SIMS investigation into tablet discolouration

Analysis by time-of-flight secondary ion mass spectrometry

Scanning electron microscopy was used to identify the cause of a glaze pinhole fault in sanitaryware manufacture

Lucideon's assistance to Pretty's Solicitors
