White Papers
View or download our white papers here. All white papers are freely available to registered users.

Lucideon has developed geopolymer formulations for diverse applications and has built the understanding to control important characteristics

This white paper introduces the most common methods for ceramic additive manufacturing
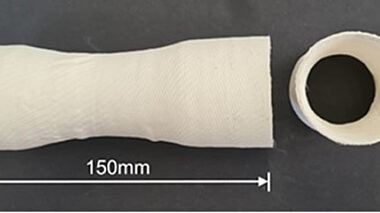
Geopolymer matrix composites are a group of materials which bridge the temperature performance gap between reinforced polymers and ceramic matrix composites
A geopolymer formulation characterized by a low initial viscosity, suitable to immobilise a wide range of waste types such as oils, chemicals and solid waste, is introduced and characterised

In this white paper, we discuss the journey behind Lucideon's proprietary iCRT-deter technology

Additive Manufacturing can provide innovative forming opportunities and can offer the potential of being able to be integrated alongside traditional methods

This white paper provides tips on what to look out for when installing refractory linings in petrochemical units

This white paper provides a broad overview of the concreting process and discusses common deficiencies and degradation mechanisms in hardened concrete structures that require petrographic examination
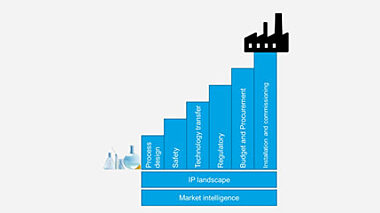
This paper gives an overview of the scale up process, highlighting possible drawbacks and best practice
Nosocomial infections (hospital acquired infections) are a major but often preventable threat to patient safety

This white paper looks at the challenges relating to materials and processing optimisation in lithium-ion batteries

This paper sets out to affirm the value of material characterisation in product and process development activities in technology based industries

This white paper discusses milling options, their advantages and disadvantages, as well as focusing on how the milling process can help to control key characteristics to optimize the properties of end products in manufacturing processes
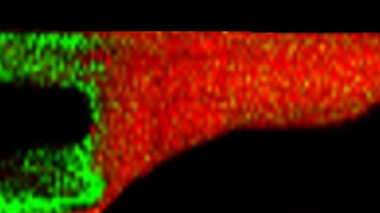
This white paper discusses the power of ToFSIMS across a range of different industries and materials and the specific information and value provided
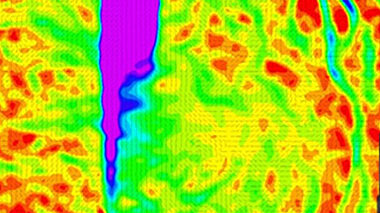
Digital Image Correlation is a technique that can deliver video footage of the strain development on the surface of a material due to loading or other actions

Drying is an important process in almost all industry sectors, including ceramics, pharmaceutical, food, chemical, construction and semiconductors

Drying is an important and often necessary unit operation for many processes, determining the quality of the final product and its shelf life

This white paper provides an introduction to the theory behind fatigue in general, and the factors that affect how materials perform in fatigue

The purpose of this paper is to show some examples where the results from whole building tests have demonstrated distinct improvements in performance

In this white paper we discuss some of the testing and validation considerations that pharmaceutical manufacturers should look at when implementing single use systems
This white paper provides some insights into the failure of plastic products and introduces some of the analytical techniques commonly used in failure diagnosis

The goal of this white paper is to demonstrate the risk of working with the wrong material, to highlight the upside when the right material is selected
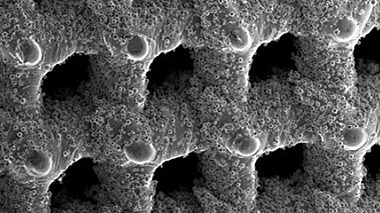
In this white paper, we will take a look at additive manufacturing from the metallurgical perspective. As with any other manufacturing process, different materials provide different benefits for particular applications
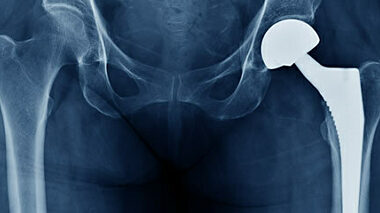
This white paper offers additional guidance that could help support implant manufacturers in their decision-making and justifications for regulatory submissions

In this white paper we discuss the sol-gel process for making ceramics and glasses
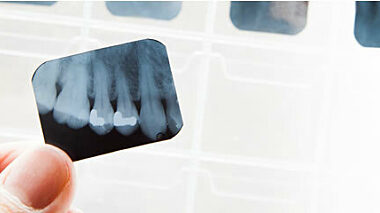
In this white paper we discuss what role inorganics can play in restoring and maintaining tooth functionality and the advantages that inorganics can provide in this area for developing technologies

Advances in high resolution imaging of both topographical features and chemical species’ distribution have been applied increasingly in the characterisation of commercially important surfaces

In this white paper, we review some of the industrial applications of surface engineering and the techniques used to define the topography and chemical composition of the surface and subsurface regions

Wear simulation for implantable knee, hip and intervertebral spinal disc prostheses have all been well documented and standardised tests methods have been created to assess the performance of these medical device implants

The applications of Digital Image Correlation are vast, from eyeball pressure testing to earthquake analysis

This white paper focuses on potential applications for Digital Image Correlation in the construction industry
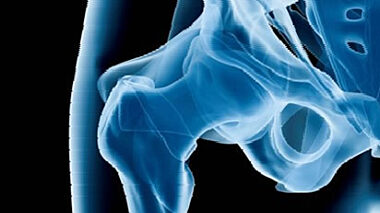
This paper will explore the applications of Digital Image Correlation technology in the healthcare sector

The concept of Quality by Design (QbD) is not a new idea but it is only in recent years that it has been considered for all aspects of the development process for pharmaceutical products
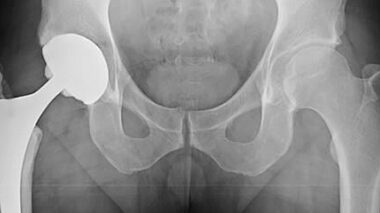
In this white paper we look at why the testing of implants is so important. Traditional test methods and their limitations are detailed and adverse event testing is discussed
This white paper explores the market drivers for abuse deterrence and identifies possible solutions

Composites are being used more and more in many different industries, thanks to the enhanced properties that are realised from the combining of materials

In this white paper we review the key tribology & testing techniques available for orthopaedic implants
The surface characterisation of hair fibres can deliver important insights into the performance of hair care products and in the development of improved product formulations

This white paper describes examples of how electron microprobe analysis has been used effectively in problem solving
This white paper discusses the applications of zeta potential for the manufacture of certain healthcare materials and how Lucideon has assisted manufacturers in this area

This white paper discusses the theory of zeta potential, and how it can be measured and controlled

In this white paper we look at the case for using one supplier for regulatory approval testing, using the example of hydroxyapatite testing
Due to the ageing UK population, increased dynamism of people's lives and growing life expectancy, there is an increasing clinical demand for bone replacement and repair
This white paper introduces the basic concepts regarding the structure and properties of polymeric materials
Much research has been done into developing synthetic Hydroxyapatite (HA) as materials for bone replacement, since natural bone comprises HA

This white paper examines the major issues involved with the investment casting process

This white paper reviews some of the technology being used in the development of new stents and how, in particular, computational modelling and material characterisation are helping to improve clinical outcomes

This white paper discusses some of the surface analysis techniques available and provides case study examples, showing where the techniques have been able to solve failure problems

Secondary Ion Mass Spectrometry (SIMS) is one of the most important characterisation tools in the semiconductor industry

Residues on the surface of medical devices can cause implant failure and poor device performance
Surface Analysis is assisting the pharmaceutical industry in a number of ways, including counterfeit detection

This white paper examines the valuable role that testing can play in relation to reducing construction costs
This white paper highlights some of the testing involved in ISO 13356 and discusses how recent and ongoing research into ceramic processing provides opportunities to meet product improvement challenges

This white paper looks at applications and potential applications of ceramic materials in the aerospace and defence industry

This white paper outlines analysis techniques for measuring concentrations of corrosion-assisting elements in nuclear reactors
