Surface Analysis Services
Lucideon specialises in surface, near surface, and interface analysis, utilising a variety of techniques to provide comprehensive testing and characterisation services for diverse applications and industries. Our expertise in surface analysis allows us to probe into microscopic features, providing insights into material properties and behaviours that impact product performance and interaction.
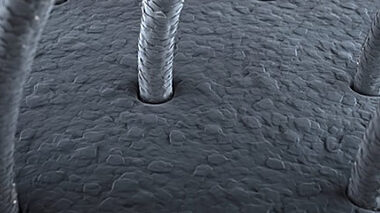
From raw materials and powders to finished products and beyond, we employ microscopy to evaluate all stages of the product life cycle, identifying manufacturing issues, changes in service, and characterising morphologies and microstructures. Through microanalysis, we can detect inclusions, stains, and deposits, and assess reactions within the end-use environment.

By measuring and visualising the chemistry and morphology of material surfaces, we can determine factors such as wettability, adhesion, biocompatibility, lubricity, colour/finish, printability, and corrosion resistance, empowering our clients with valuable insights into their materials and products.
Our world-leading surface science facilities allow characterisation of elements and their isotopes, chemical groups, polymers, and surface additives. Surface analysis techniques allow us to measure elemental and molecular chemistry of all elements from H to U in the ppm - ppb range, and perform materials characterisation on any material, including drugs.
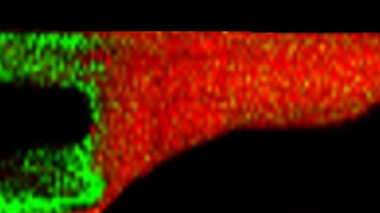
Elemental and molecular imaging can be used to map across an outer surface or cross-section of a material or device.
Surfaces can be analysed to identify the presence of dopants, process contaminants, impurities, and surface treatments to depths of tens of microns. Depth analysis allows characterisation of layer structures, buried features, and surface treatments. Multi-element depth profiling can be performed to depths from nm to tens of µm.
Typical applications include profiling semiconductor device features, compositional analysis of glass layer structures, investigation of stains and rinse residues, monitoring of surface cleaning treatments, characterising changes in polymer functionality following plasma treatments, identifying contaminants, solving adhesive/bonding issues, and in-depth investigations of tribological surfaces, including wear scars.
Non-destructive testing can be performed so samples don’t have to be sacrificed.
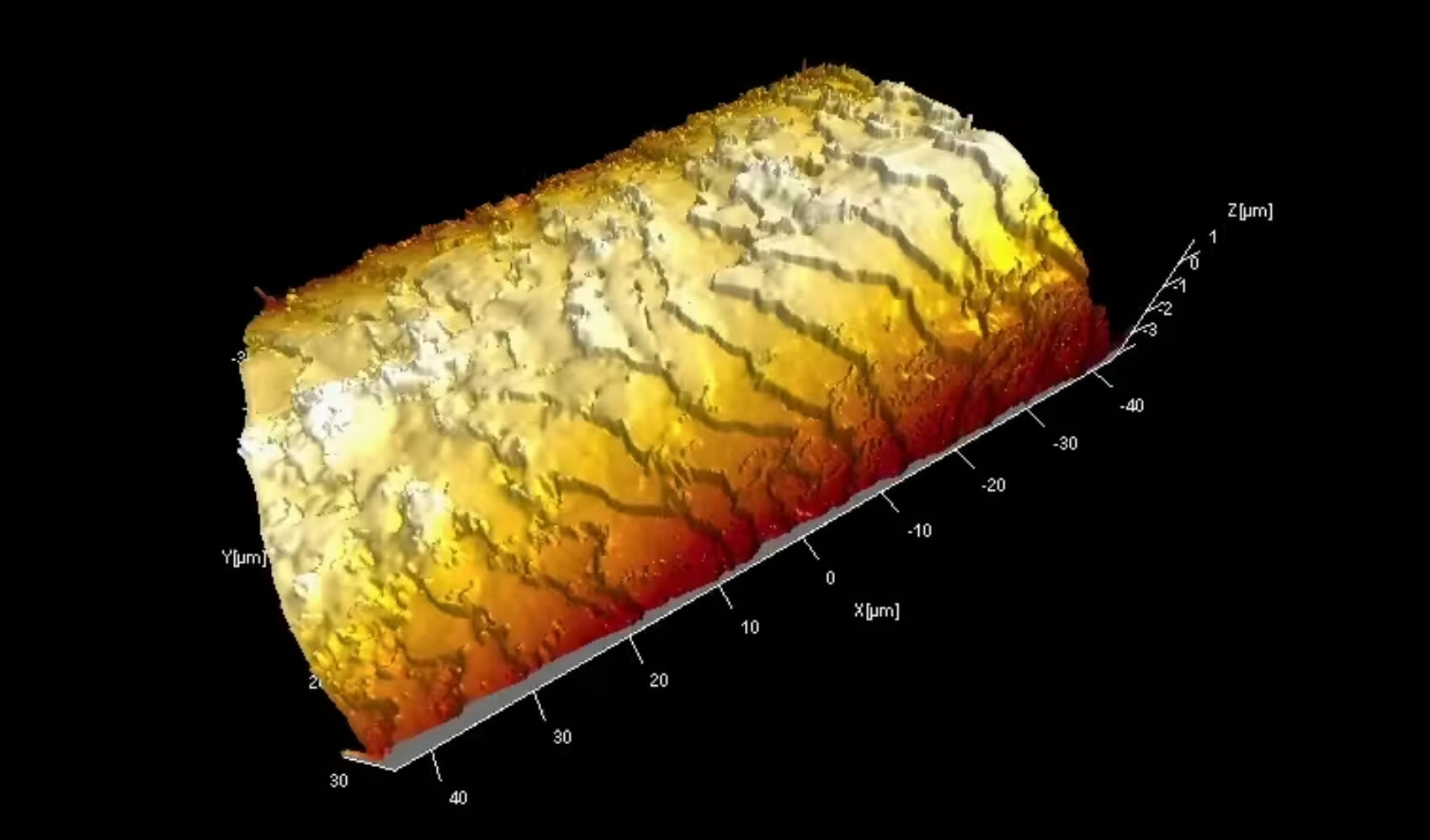
Surface analysis techniques are compatible with retrospective 3D mapping techniques to show chemical maps and depth profiles. Chemical mapping can show features down to <1 micron in size, a useful technique for the clear diagnosis of fault areas, among other applications. Surface Topography is used to generate 3D maps on nm scale showing microstructure, wear, etc. and can also be applied to cross-sectional materials to give extended information through a structure, which is particularly useful for complex, thick layered materials.
3D Visualisation of human hair following conditioner treatments
The video clip below depicts the 3D visualisation of a human hair surface, by white light interferometry, measuring the surface topography and roughness after shampoo-conditioner treatments.
Analytical techniques for surface analysis include:
- XPS (X-ray Photoelectron Spectroscopy)
- ToF-SIMS (Time-of-Flight Secondary Ion Mass Spectrometry)
- WLI (White Light Interferometry)
- SEM/EDX (Scanning Electron Microscopy / Energy Dispersive X-ray Analysis)
- 3DSEM (Three Dimensional Scanning Electron Microscopy)
- XRD (X-Ray Diffraction)
- FTIR (Fourier Transform Infrared Analysis)
- AFM (Atomic Force Microscopy)
- TEM (Transmission Electron Microscopy)
- Raman Spectroscopy
White Papers